Cookie Settings
We use cookies to provide an optimal experience for you. Technically required cookies are used for making shopping possible, statistics are used for anonymized Google Analytics. You can read everything in our updated privacy policy.
| Sample Material | 50ml EDTA whole blood |
| Insulation method | Non-magnetic with 300 µl CD19 S-pluriBeads® anti-human |
| Yield | ~3.5 * 10^6 B lymphocytes |
| Vitality | >99% (Trypan blue stain) |
| Purity determination | Anti-CD20 FITC antibody staining followed by FACS analysis. |
| Purity | ~97% B lymphocytes |
After target activation, the abundance of antibody-secreting plasma cells (ASC) was determined by ELISpot.
Isolated B cells were stimulated with a polyclonal activation mixture for 5 days. They were then grown on a plate coated with the specific antigen. After 24 h incubation, the assay was performed.
The spots were counted and evaluated with a special software (AID ELISpot6.0 iSpot). In the adjacent diagram, the blue numbers indicate the number of antigen-specific spots per well.
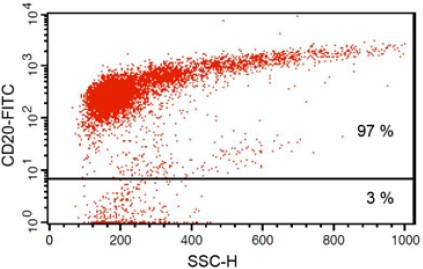
Fig 1: Purity of isolated & stimulated B lymphocytes
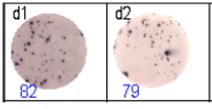
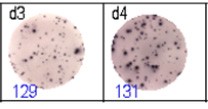
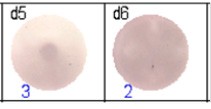
Fig 2: ELISpot analysis of anti-Ig specific cells in duplicates. d1d2: 250 cells/well each, d3d4: 500 cells/well each, d5d6: non-activated cells (500 cells/well each)
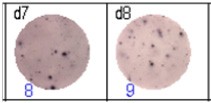
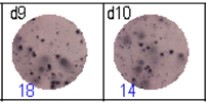
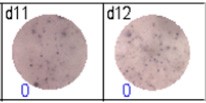
Fig 3: d7d8: Anti-influenza nucleocapsid-specific memory B cells, d9d10: nti tetanus toxoid-specific memory B cells, d11d12: negative control.
S-pluriBead® CD19 enabled the isolation of B lymphocytes from a human whole blood sample with a purity of ~ 97%.
The isolated targets showed >99% viability after trypan blue staining. They were successfully grown in cell culture as well as differentiated into antibody-secreting plasma cells (ASCs). ASCs were found to be suitable for analysis of antigen-specific memory B cell response by ELISpot assay. The results were comparable to those obtained with a reference method (Dynabeads® CD19 Pan B).
For the reference method, see publication: Bussmann BM, Reiche S, Bieniek B, Krznaric I, Ackermann F, Jassoy C: Loss of HIV-specific memory B-cells as a potential mechanism for the dysfunction of he humoral immune response against HIV. Virology 2010; 397(1):7-13.)